Degarelix, sold under the brand name Firmagon, is a pivotal hormonal therapy in the management of prostate cancer. As a synthetic peptide and a potent Gonadotropin-Releasing Hormone (GnRH) receptor antagonist, its primary and most well-established use is for androgen deprivation therapy (ADT) in patients with advanced, hormone-sensitive prostate cancer. Unlike traditional GnRH agonists, Degarelix offers a unique mechanism of action that provides rapid and sustained testosterone suppression without an initial tumor-stimulating flare, making it a critical treatment option for specific patient populations. This article explores its core indications, mechanism, clinical efficacy, safety profile, and emerging applications, supported by authoritative clinical data and regulatory guidelines.
Mechanism of Action: Direct and Rapid Hormonal Suppression
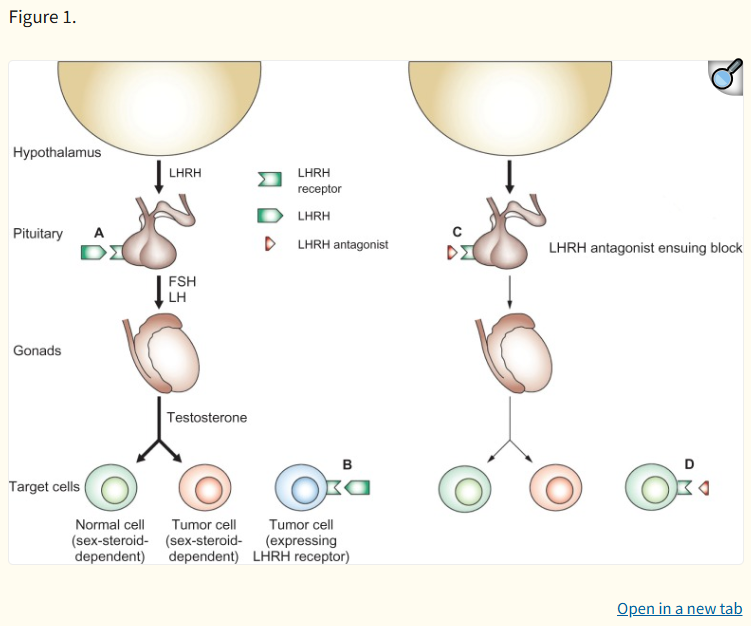
The therapeutic power of Degarelix stems from its direct competitive blockade of GnRH receptors in the pituitary gland. The hypothalamus naturally releases GnRH to stimulate the pituitary to secrete Luteinizing Hormone (LH) and Follicle-Stimulating Hormone (FSH). These hormones then signal the testes to produce testosterone, a key driver of prostate cancer cell growth.
Degarelix binds tightly and reversibly to these pituitary receptors, immediately halting the release of LH and FSH. This swift inhibition leads to a dramatic and rapid reduction in testicular testosterone production. A defining advantage over GnRH agonists is the absence of an initial "testosterone surge" or clinical flare. Agonists initially overstimulate the receptors, causing a transient spike in testosterone that can worsen symptoms like bone pain or urinary obstruction in vulnerable patients. Degarelix avoids this dangerous phase, beginning testosterone suppression on the first day of administration and achieving medical castration levels (≤0.5 ng/mL or 50 ng/dL) in over 96% of patients by day 3. This rapid onset is crucial for patients requiring immediate disease control.

Primary Approved Indications: Advanced and High-Risk Prostate Cancer
Degarelix is globally approved, including by the U.S. FDA and the European Medicines Agency (EMA), specifically for the treatment of advanced, hormone-dependent prostate cancer. Its clinical profile makes it particularly suited for several key scenarios within this broad indication:
Advanced Prostate Cancer (Metastatic or Locally Advanced): This is the core FDA-approved use. It is indicated for patients with metastatic disease, where cancer has spread beyond the prostate, or locally advanced disease (T3/T4), where it has spread outside the prostate but not to distant organs. The goal is to induce and maintain long-term testosterone suppression to slow or halt cancer growth.
High-Risk Localized Prostate Cancer (in Combination with Radiotherapy): The EMA and clinical guidelines approve Degarelix as part of a combined modality approach. For patients with high-risk localized or locally advanced prostate cancer, it is used as neoadjuvant (pre-treatment) and concurrent therapy with radiation therapy. This combination improves local control and survival by shrinking the tumor before radiation and suppressing hormone-driven growth during treatment.
Patients at Risk of Flare Symptoms: Due to its lack of initial testosterone surge, Degarelix is the preferred ADT choice for patients with symptomatic metastases, impending spinal cord compression, severe urinary obstruction, or those at high cardiovascular risk. These patients cannot tolerate the potential symptom exacerbation caused by GnRH agonists.
Second-Line Therapy or for Patients Intolerant to Agonists: It serves as an effective alternative for patients who have progressed on or cannot tolerate the side effects of standard GnRH agonist therapies.

Clinical Efficacy: Proven Performance in Key Trials
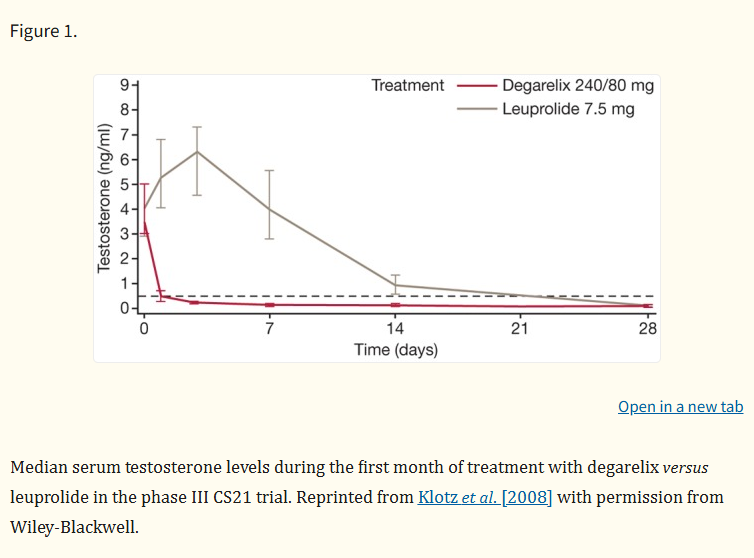
The efficacy of Degarelix has been rigorously established in Phase III clinical trials, most notably a large, randomized, open-label study comparing it to the GnRH agonist leuprolide in over 600 prostate cancer patients.
The primary endpoint was maintaining testosterone at castrate levels for one year. The results demonstrated that Degarelix (at both 80mg and 160mg maintenance doses) was non-inferior to leuprolide, with over 97% of patients achieving sustained suppression. The trial's most striking finding was the speed of suppression: by day 3, 96.5% of Degarelix patients were castrated, compared to 0% in the leuprolide group. This rapid control was accompanied by a significantly faster decline in Prostate-Specific Antigen (PSA) levels, a key biomarker for disease activity.
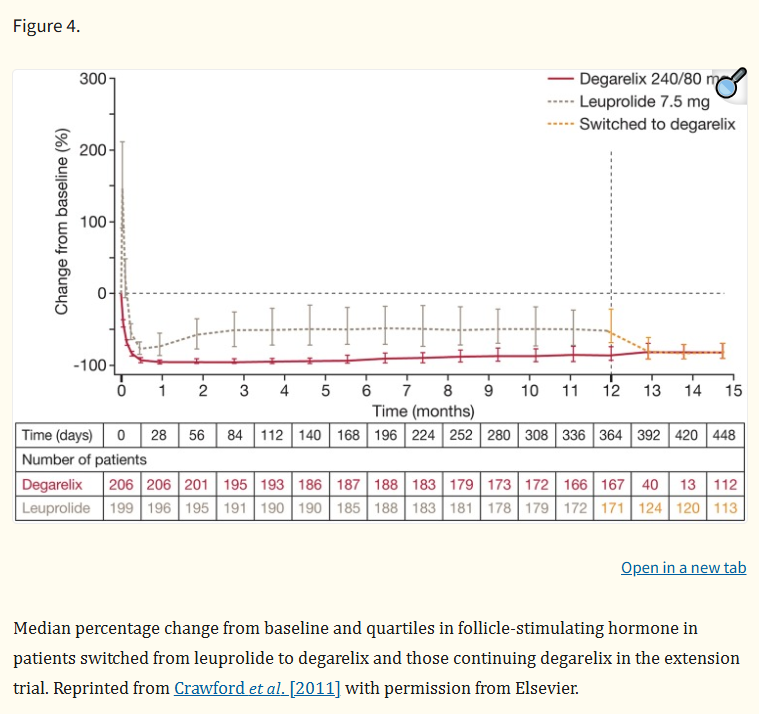
A comprehensive meta-analysis confirmed these findings, further highlighting that Degarelix provides superior relief from lower urinary tract symptoms and is associated with a lower risk of cardiovascular adverse events compared to agonists. Its efficacy is maintained with monthly subcutaneous injections, forming a convenient and effective long-term treatment regimen.
Safety Profile and Administration
Degarelix is administered as a subcutaneous abdominal injection, with a starting dose of 240mg (two 120mg injections) followed by monthly maintenance doses of 80mg. Its safety profile is well-characterized.
The most common side effects are injection-site reactions (e.g., pain, redness, swelling), occurring in about 40-50% of patients, primarily with the initial dose. These are generally mild-to-moderate and decrease over time. Other frequent side effects are typical of ADT and include hot flashes, weight changes, fatigue, and transient increases in liver enzymes (transaminases).
A significant safety advantage is its more favorable cardiovascular profile compared to GnRH agonists. Meta-analyses show a lower incidence of major adverse cardiovascular events, a critical consideration for older patients with multiple comorbidities. However, as with all ADT, healthcare providers must monitor for potential QT interval prolongation, especially in patients with pre-existing heart conditions or on concomitant medications that affect the heart's electrical cycle.Degarelix is contraindicated in patients with severe hypersensitivity to the drug and in women who are or may become pregnant.
Emerging and Off-Label Applications
While advanced prostate cancer remains its only formal indication, research is exploring Degarelix's potential in other areas. Its potent and rapid testosterone suppression is being investigated for neoadjuvant therapy prior to radical prostatectomy in high-risk patients, aiming to shrink tumors and improve surgical outcomes. Preliminary studies also suggest a potential role in managing castration-resistant prostate cancer (CRPC) in combination with newer androgen receptor-targeted therapies.

There is also limited research into its use for benign conditions like benign prostatic hyperplasia (BPH), where reducing testosterone can alleviate urinary symptoms, although this remains off-label and not a standard of care.
Conclusion
In summary, Degarelix (Firmagon) is a specialized and highly effective GnRH antagonist, primarily used for the androgen deprivation therapy of advanced, hormone-sensitive prostate cancer. Its unique selling points are its rapid onset of action, absence of a testosterone flare, and favorable cardiovascular safety profile relative to older agonist therapies. These characteristics make it the treatment of choice for patients needing immediate disease control or those at risk of complications from a hormone surge. Backed by robust clinical trial data, Degarelix represents a significant advancement in hormonal therapy, offering a valuable and often preferred option for urologists treating patients across the spectrum of advanced and high-risk prostate cancer.
Post time: 2026-04-13
